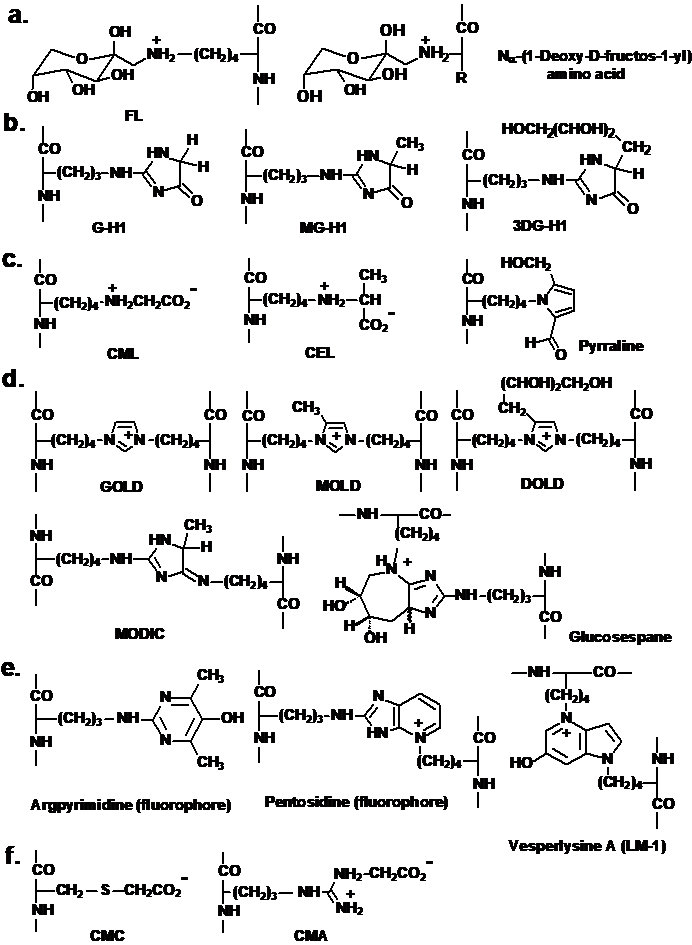Glycation – nomenclature and classification
In 1985 the Nomenclature Committee of International Union of Biochemistry and the International Union of Pure and Applied Chemistry recommended the term glycation for “all reactions that link a sugar to a protein or peptide, whether or not catalyzed by an enzyme” – drafted by Sharon (Sharon, 1986). In a subsequent review in 1993, Lis and Sharon distinguished between enzymatic modification of proteins by saccharides in processes of protein glycosylation forming glycoproteins and non-enzymatic modification of proteins by saccharides in processes of protein glycation forming glycated proteins (Lis and Sharon, 1993). This eventually led to selective use of the term glycation for non-enzymatic modification of proteins by saccharides such that it is now accepted nomenclature. In older scientific literature, one of the most prominent glycated proteins, hemoglobin, has been called glycosylated hemoglobin, non-enzymatic glycosylated haemoglobin, glycohemoglobin, glucosylated hemoglobin and other variants.
Protein glycation involves the non-enzymatic attachment of a reducing sugar or sugar derivative to a protein. The term “reducing” related to the classical activity of saccharides with solution species with free aldehyde or ketone groups which reduced Benedict’s solution (Benedict, 1909). Glycation is not available to non-reducing oligosaccharides where aldehyde or ketone groups of component monosaccharides have been converted to ketal and acetal groups of glycosidic bonds – such as in sucrose (table sugar). In its earliest development, protein glycation was thought to be restricted to modification of amino groups of lysine residue side chains N-terminal amino acid residues. In more recent times, glycation of arginine residues by dicarbonyl metabolites has emerged as a major feature of protein glycation in physiological systems. There is also involvement of cysteine residues.
Glycation of proteins occurs by a complex series of sequential and parallel reactions called collectively the Maillard reaction – named after the leading pioneer of glycation research, Louis Camille Maillard (1978 – 1936). Many different adducts may be formed – some of which are fluorescent and coloured “browning pigments”. In the physiological setting, one of the important saccharides participating in glycation of mammalian metabolism is glucose, and some of the most important saccharide derivatives are the reactive dicarbonyl metabolites such as methylglyoxal (MG).
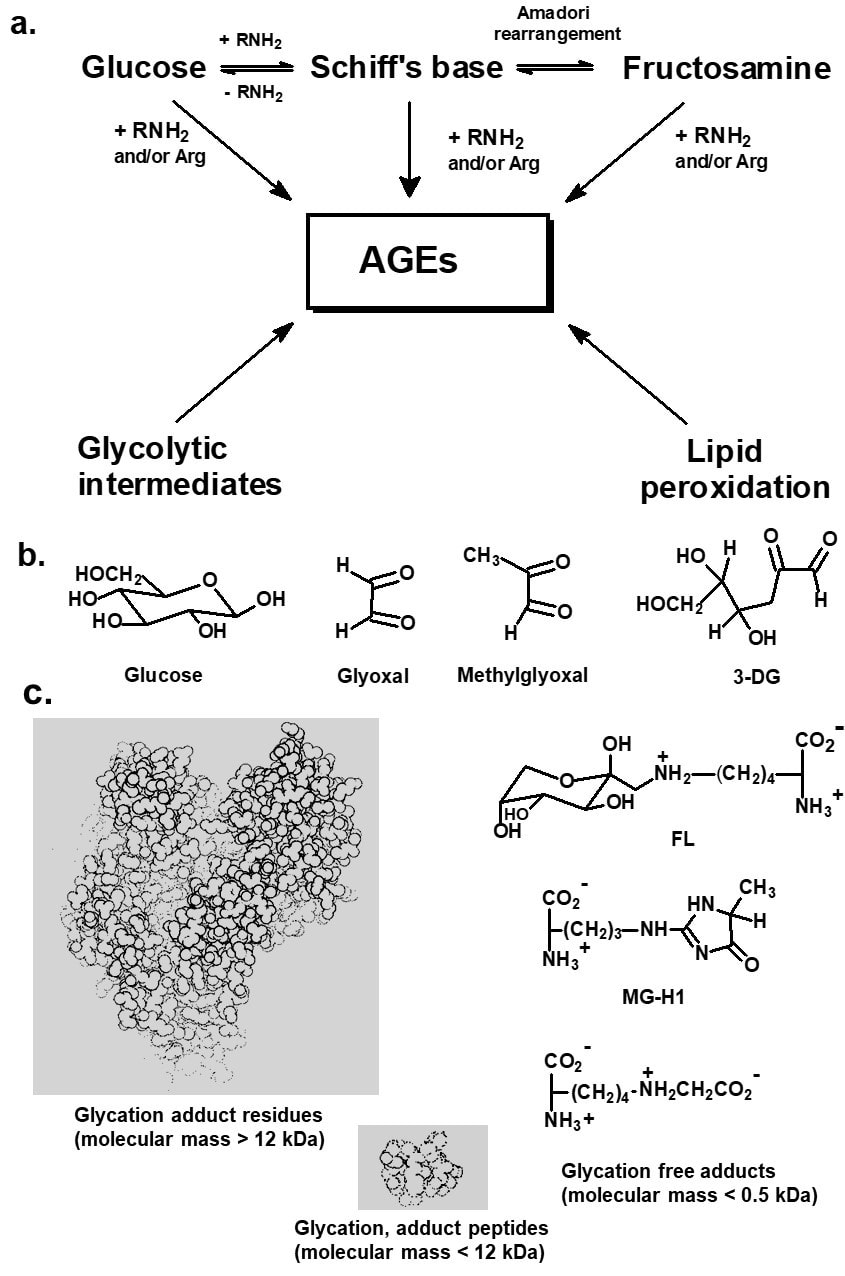
Historically, glycation of lysyl side chain and N-terminal amino groups by glucose was the major glycation process studied in the glycation of proteins. This has given rise to the classification of early and advanced glycation processes. Glucose reacts with amino groups to initially form a glycosylamine which dehydrates to form a Schiff’s base. The Schiff’s base undergoes an Amadori rearrangement to form N-(1-deoxy-D-fructos-1-yl)amino acids or fructosamine (Hodge, 1955). Glycation of proteins by glucose to the fructosamine stage has been classified as early glycation. Glycoslyamine, Schiff’s base and fructosamines are considered to be early stage glycation adducts. In later stage reactions fructosamine degrades to many stable end-stage adducts called advanced glycation endproducts (AGEs) – Figure 1a.
Protein glycation involves the non-enzymatic attachment of a reducing sugar or sugar derivative to a protein. The term “reducing” related to the classical activity of saccharides with solution species with free aldehyde or ketone groups which reduced Benedict’s solution (Benedict, 1909). Glycation is not available to non-reducing oligosaccharides where aldehyde or ketone groups of component monosaccharides have been converted to ketal and acetal groups of glycosidic bonds – such as in sucrose (table sugar). In its earliest development, protein glycation was thought to be restricted to modification of amino groups of lysine residue side chains N-terminal amino acid residues. In more recent times, glycation of arginine residues by dicarbonyl metabolites has emerged as a major feature of protein glycation in physiological systems. There is also involvement of cysteine residues.
Glycation of proteins occurs by a complex series of sequential and parallel reactions called collectively the Maillard reaction – named after the leading pioneer of glycation research, Louis Camille Maillard (1978 – 1936). Many different adducts may be formed – some of which are fluorescent and coloured “browning pigments”. In the physiological setting, one of the important saccharides participating in glycation of mammalian metabolism is glucose, and some of the most important saccharide derivatives are the reactive dicarbonyl metabolites such as methylglyoxal (MG).
Historically, glycation of lysyl side chain and N-terminal amino groups by glucose was the major glycation process studied in the glycation of proteins. This has given rise to the classification of early and advanced glycation processes. Glucose reacts with amino groups to initially form a glycosylamine which dehydrates to form a Schiff’s base. The Schiff’s base undergoes an Amadori rearrangement to form N-(1-deoxy-D-fructos-1-yl)amino acids or fructosamine (Hodge, 1955). Glycation of proteins by glucose to the fructosamine stage has been classified as early glycation. Glycoslyamine, Schiff’s base and fructosamines are considered to be early stage glycation adducts. In later stage reactions fructosamine degrades to many stable end-stage adducts called advanced glycation endproducts (AGEs) – Figure 1a.
Figure 1. Protein glycation. (a) Early glycation and sources of advanced glycation endproducts (AGEs). b. Some important glycating agents. c. Classification of protein glycation adduct residues, peptides and free adducts. The 12 kDa discrimination for protein glycation adduct residues and peptides is empirical and relates to the pore size of ultrafiltration membrane used in separation.
This is a broader definition of AGEs than originally given by Cerami as “brown fluorescent pigments which crosslink proteins” (Cerami, 1986). Inconsistencies in the application of this classification and nomenclature have arisen because glucose may degrade when unattached to protein to form reactive ɑ-oxoaldehydes. ɑ-Oxoaldehydes are potent glycating agents and react with proteins to form AGEs directly. The Schiff’s base adduct may also degrade via non-Amadori rearrangement reaction pathways to ɑ-oxoaldehydes which also leads to the formation of AGEs. The latter are also formed by the direct modification of proteins by ɑ-oxoaldehydes produced by the degradation of glycolytic intermediates and lipid peroxidation. Important ɑ-oxoaldehyde or dicarbonyl glycating agents are glyoxal, MG, and 3-deoxyglucone (3-DG) – Figure 1b. Therefore, AGEs may be formed in glycation processes by glucose in pre- and post-Amadori product reactions, and indeed in processes where an Amadori product is not a precursor. AGEs may be formed in both the early and late stages of glycation processes. The term “advanced glycation endproduct” is a misnomer in that AGEs are formed in both early and later, advanced stages of the Maillard reaction. The concept of early and advanced glycation adducts is, however, simple and the nomenclature of fructosamine and AGEs is convenient, widely used, understood and accepted. Molecular structures of fructosamines and some common AGEs are given in Figure 2.
Figure 2. Molecular structures of glycation adduct residues. a. Early glycation adducts formed by glycation with glucose: fructosamines. R indicates the amino acid side chain. b. – f. Advanced glycation endproducts. b. Hydroimidazolones. c. Monolysine adducts. d. imidazolium and other crosslinks. e. fluorophores, and f. other structures. The peptide linkage is shown cut away for clarity. For the corresponding free adducts at physiological pH, the N-terminal amino group is protonated –NH3+ and the C-terminal carbonyl is a carboxylate –CO2- moiety. Ionisation status is given for the major solution form at pH 7.4.
Further classification of AGEs has been made to relate to the mechanism of AGE formation. “Glycoxidation” is a term in general use for glycation processes in which oxidation is involved and the AGEs formed thereby have been called “glycoxidation products” (Baynes, 1991). The AGEs pentosidine and CML are examples of glycoxidation products. Where aldehyde substrates of glycation are formed by lipid peroxidation, the end stage adducts have been called advanced lipoxidation adducts (ALEs). CML is also an ALE as it maybe be formed from glyoxal generated from lipid peroxidation (Fu et al., 1996) Other classifications were devised but currently are not in widespread use (Baynes and Thorpe, 2000) – Figure 1.
Glycation adducts of proteins are formed by modification of amino acid residues of proteins with monosaccharides or saccharide derivatives. They are appropriately called, therefore, glycation adduct residues – such as the fructosamine residues of glycated hemoglobin and AGE residues of AGE-modified proteins. Glycated proteins undergo proteolysis during digestion of ingested proteins in food and turnover of proteins in tissues and body fluids. Proteolysis of glycated proteins forms lower molecular mass glycated peptides and glycated amino acids. The latter are conveniently called glycation free adducts. Glycation free adducts are acutely sensitive to change in renal clearance in mammalian systems (Rabbani et al., 2007) – Figure 1c.
Glycation adduct residues of proteins have been called “protein-bound” glycation adducts - particularly for AGEs in renal failure research. This is discouraged as it is a misnomer; part of the glycation adduct structure is an integral part of the protein and hence binding of discrete glycation adduct is not involved. There may be non-covalent binding of discrete binding of glycation free adducts to plasma protein in some instances, particularly at the high levels of glycation free adducts found in renal failure (Agalou et al., 2005,Rabbani et al., 2007) and hence it is important to be able to distinguish between glycation adduct residues and binding of free adducts.
Glycation adducts of proteins are formed by modification of amino acid residues of proteins with monosaccharides or saccharide derivatives. They are appropriately called, therefore, glycation adduct residues – such as the fructosamine residues of glycated hemoglobin and AGE residues of AGE-modified proteins. Glycated proteins undergo proteolysis during digestion of ingested proteins in food and turnover of proteins in tissues and body fluids. Proteolysis of glycated proteins forms lower molecular mass glycated peptides and glycated amino acids. The latter are conveniently called glycation free adducts. Glycation free adducts are acutely sensitive to change in renal clearance in mammalian systems (Rabbani et al., 2007) – Figure 1c.
Glycation adduct residues of proteins have been called “protein-bound” glycation adducts - particularly for AGEs in renal failure research. This is discouraged as it is a misnomer; part of the glycation adduct structure is an integral part of the protein and hence binding of discrete glycation adduct is not involved. There may be non-covalent binding of discrete binding of glycation free adducts to plasma protein in some instances, particularly at the high levels of glycation free adducts found in renal failure (Agalou et al., 2005,Rabbani et al., 2007) and hence it is important to be able to distinguish between glycation adduct residues and binding of free adducts.
Historical background of glycation
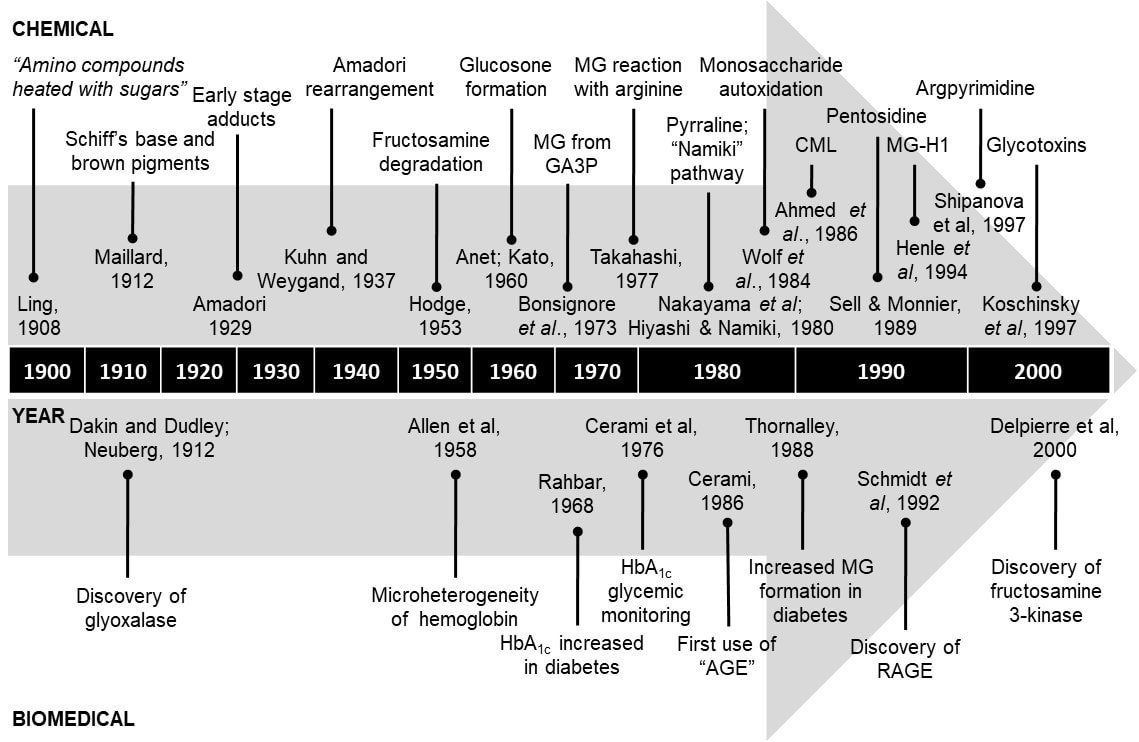
In 1908, Arthur Robert Ling (1861 – 1937) produced flavoured and coloured compounds from thermal drying of proteins with sugars. He wrote “When these amino-compounds produced from proteins are heated at 120 - 140°C with sugars such as ordinary glucose or maltose, which are produced at this stage of process, combination occurs. The precise nature of the compounds produced is unknown to me, but they are probably glucosamine - like bodies" (Ling, 1908). He also described the heating of glucose with asparagine producing browning pigments. These were amongst the earliest recorded adducts formed between glucose and an amino acid. In 1912, Louis Camille Maillard investigated the reaction between glucose and glycine on heating. He proposed that the brown pigmented product or melanoids involved an initial interaction between amines and saccharides forming Schiff’s base adducts (Maillard, 1912a). The non-enzymatic reaction of amino acids with sugars now bears his name: “Maillard reaction”. Maillard acknowledged the prior work of Ling in a later paper (Maillard, 1912b). In 1913 the glyoxalase system that catalyses the conversion of MG to lactate and thereby suppresses dicarbonyl glycation in physiological systems was discovered (Dakin and Dudley, 1913,Neuberg, 1913). In 1925 and 1931, Mario Amadori demonstrated that the condensation of D-glucose with aromatic amines p-phenetidine, p-anisidine or p-toluidine gave two structurally different isomers which were not anomers (Amadori, 1929a,Amadori, 1929b). One of the isomers was more labile than the other towards hydrolysis and was also more susceptible to decomposition on standing in the solid state in air. He correctly recognised this as the N-glycosylamine but he mistakenly thought that the more stable isomer was a Schiff’s base, overlooking its resistance to acid hydrolysis(Amadori, 1929a). In 1936, Kuhn and Dansi found that the stable isomer was not a Schiff’s base but rather the product of a molecular rearrangement. They also confirmed that the labile isomer was in fact the N-substituted glycosylamine (Kuhn and Dansi, 1936). In 1937, Kuhn and Weygand reported the structure of the Amadori’s stable isomer, which was the unbranched N-substituted 1-amino-1-deoxy-2-ketose. Kuhn and Weygand later called the reaction the Amadori rearrangement involving generally aldoses and amines (Kuhn and Weygand, 1937). In 1953, Hodge suggested that the Maillard reaction involved formation of a Schiff’s base followed by Amadori rearrangement in the early stages. He also suggested that fructosamine degraded to glucosone and other adducts via enolization, oxidation and fragmentation reactions (Hodge, 1953). An excellent history of the early days of glycation research was given by Kawamura (Kawamura, 1983).
In 1958, Allan et al. reported the first example of a glycated protein in vivo. They reported the presence of a negatively charged component of haemoglobin in human red blood cells (Allen et al., 1958). This was variants of glycated haemoglobin HbA1. It was first characterized as a glycated hemoglobin by Bookchin and Gallop in 1968 (Bookchin and Gallop, 1968), its increase in diabetes described by Rahbar in 1968 (Rahbar, 1968), reactions leading to its formation were characterized by Bunn and his co-workers in 1975 (Bunn et al., 1975) and the use of the glucose adduct glycated hemoglobin HbA1c for monitoring the glycemic control in diabetic patients was proposed by Anthony Cerami and co-workers in 1976 (Koenig et al., 1976). Percentage of HbA1c is now a routine clinical diagnostic measure of glycaemic control of diabetic patients worldwide.
In 1960, Anet reported the degradation of a fructosamine (N,N-difructosylglycine) to 3-deoxyglucosone (Anet, 1960). This was the first example of the importance of fructosamine dehydration to a 3-deoxy-2-oxoaldehyde. Kato also isolated 3-deoxyglucosone and 3-deoxypentosone from the browning reactions of glucose and ribose with an amine (Kato, 1960,Kato et al., 1969,Kato et al., 1989). These reactive ɑ-oxoaldehyde products of the Maillard reaction are now seen as important precursors of glycation adduct formation in biological systems.
In 1973, Bonsignore et al. presented the first evidence that a triosephosphate, glyceraldehyde-3-phosphate, degraded non-enzymatically under physiological conditions to form MG (Bonsignore et al., 1973). In 1977, Takahashi described the reaction of amino acids with glyoxal derivatives, including glyoxal and MG. Arginine was identified as the predominant amino acid modified and a hydroimidazolone was one molecular structure proposed for the adducts (Takahashi, 1977). Although no supporting analytical data were given, this was later to be confirmed as a major AGE in physiological systems and some foodstuffs (Ahmed et al., 2002,Henle et al., 1994,Thornalley et al., 2003).
In 1980, Nakayama et al. reported the formation of 6-(2-formyl-5-hydroxymethylpyrrol-1-yl)-L-norleucine from 3-deoxyglucosone and lysyl residues in proteins, now commonly known as pyrraline - an AGE (Nakayama et al., 1980). Hayashi and Namiki presented evidence for the fragmentation of the saccharide moiety early in the Maillard reaction, leading to the formation of ɑ-oxoaldehydes. Evidence for the formation of glyoxal and MG established saccharide fragmentation as a new series of reaction pathways in Maillard chemistry, now collectively called the Namiki pathway (Hayashi and Namiki, 1980).
In 1984, a product formed from the degradation of Amadori product, 2-(2-furoyl)-4(5)-(2-furanyl)-1H-imidazole (FFI), was reported by Pongor et al. (Pongor et al., 1984). Wolff and co-workers reported the slow oxidative degradation of monosaccharides under physiological conditions to form the corresponding ɑ-oxoaldehyde and hydrogen peroxide. This process was called monosaccharide autoxidation (Wolff et al., 1984).
In 1986, Cerami first used the term advanced glycation endproducts (AGEs) to refer to “brown fluorescent pigments which crosslink proteins” formed from the degradation of fructosamine (Cerami, 1986). Brownlee et al. reported increased collagen cross-linking in the arterial walls of diabetic rats and associated fluorescence characteristic of AGE compounds. The nucleophilic hydrazine derivative, aminoguanidine (PimagedineTM), inhibited the formation of AGEs and diabetes-induced protein crosslinking (Brownlee et al., 1986). Ahmed et al. also reported the formation of Nɛ-carboxymethyl-lysine (CML) from the oxidative degradation of glycated proteins and also noticed that these two compounds were present in human urinary metabolite (Ahmed et al., 1986). CML was later found to be formed by the reaction of lysine residues with ascorbate and glyoxal formed in lipid peroxidation and is now one of the most studied AGEs in glycation research (Thorpe and Baynes, 2002).
In 1988, Thornalley presented evidence linking hyperglycaemia in diabetes mellitus with increased flux of formation and concentration of MG (Thornalley, 1988). Glycation by MG was later to be proposed as one of the key pathways driving the development of vascular complications of diabetes (Brownlee, 2001).
In 1989, Monnier and Sell isolated an acid stable fluorescent compound from collagen – “pentosidine”. It was a crosslink formed from a pentose moiety with lysine and arginine residues and is a further highly studied AGE (Sell and Monnier, 1989). In 1991, the enzymatic metabolism of fructosamine adducts was also discovered by Horiuchi and Kyurokawa - fructosyl-amino acid oxidase from bacteria catalysed the conversion of fructosyl-amino acids to the free amino acid, glucosone and hydrogen peroxide (Horiuchi and Kurokawa, 1991).
In 1992, Schmidt and co-workers identified an AGE receptor protein from bovine lung endothelial cells – later shown to have a sequence mass of 42 kDa (Schmidt et al., 1992). This was the Receptor for Advanced Glycosylation Endproducts (RAGE) and is the best characterised of the AGE receptors.
Over the period 1994 - 1998 the inhibitor of AGE formation PimagedineTM was evaluated in clinical trials for the prevention of overt nephropathy in clinical diabetes (ACTION I and ACTION II). The trials were eventually terminated due to safety concerns and apparent lack of efficacy (Thornalley, 2003c).
In 1996, Cerami and co-workers introduced the concept of “AGE breakers” – compounds that may cleave glycation derived crosslinks and reverse one of the damaging effects of glycation associated with ageing and disease. The prototype compound was N-phenacylthiazolium bromide (PTB) (Vasan et al., 1996). Unfortunately it and related analogues had stability problems (Price et al., 2001,Thornalley and Minhas, 1999).
In 1997, Shipanova et al. discovered argpyrimidine – a major fluorescence AGE formed from MG (Shipanova et al., 1997). Vlassara and co-workers used the term “glycotoxins” to refer to highly reactive AGE intermediates and initiated a series of on-going studies assessing the physiological effects of AGEs in the diet on health (Koschinsky et al., 1997).
In 2000, Delpierre et al. discovered fructosamine 3-kinase which catalyses the phosphorylation of fructosamine and fructosamine residues in proteins leading to the de-glycation and repair of early glycated proteins (Delpierre et al., 2000). A time-line diagram of glycation research development is given in Figure 3.
In 1958, Allan et al. reported the first example of a glycated protein in vivo. They reported the presence of a negatively charged component of haemoglobin in human red blood cells (Allen et al., 1958). This was variants of glycated haemoglobin HbA1. It was first characterized as a glycated hemoglobin by Bookchin and Gallop in 1968 (Bookchin and Gallop, 1968), its increase in diabetes described by Rahbar in 1968 (Rahbar, 1968), reactions leading to its formation were characterized by Bunn and his co-workers in 1975 (Bunn et al., 1975) and the use of the glucose adduct glycated hemoglobin HbA1c for monitoring the glycemic control in diabetic patients was proposed by Anthony Cerami and co-workers in 1976 (Koenig et al., 1976). Percentage of HbA1c is now a routine clinical diagnostic measure of glycaemic control of diabetic patients worldwide.
In 1960, Anet reported the degradation of a fructosamine (N,N-difructosylglycine) to 3-deoxyglucosone (Anet, 1960). This was the first example of the importance of fructosamine dehydration to a 3-deoxy-2-oxoaldehyde. Kato also isolated 3-deoxyglucosone and 3-deoxypentosone from the browning reactions of glucose and ribose with an amine (Kato, 1960,Kato et al., 1969,Kato et al., 1989). These reactive ɑ-oxoaldehyde products of the Maillard reaction are now seen as important precursors of glycation adduct formation in biological systems.
In 1973, Bonsignore et al. presented the first evidence that a triosephosphate, glyceraldehyde-3-phosphate, degraded non-enzymatically under physiological conditions to form MG (Bonsignore et al., 1973). In 1977, Takahashi described the reaction of amino acids with glyoxal derivatives, including glyoxal and MG. Arginine was identified as the predominant amino acid modified and a hydroimidazolone was one molecular structure proposed for the adducts (Takahashi, 1977). Although no supporting analytical data were given, this was later to be confirmed as a major AGE in physiological systems and some foodstuffs (Ahmed et al., 2002,Henle et al., 1994,Thornalley et al., 2003).
In 1980, Nakayama et al. reported the formation of 6-(2-formyl-5-hydroxymethylpyrrol-1-yl)-L-norleucine from 3-deoxyglucosone and lysyl residues in proteins, now commonly known as pyrraline - an AGE (Nakayama et al., 1980). Hayashi and Namiki presented evidence for the fragmentation of the saccharide moiety early in the Maillard reaction, leading to the formation of ɑ-oxoaldehydes. Evidence for the formation of glyoxal and MG established saccharide fragmentation as a new series of reaction pathways in Maillard chemistry, now collectively called the Namiki pathway (Hayashi and Namiki, 1980).
In 1984, a product formed from the degradation of Amadori product, 2-(2-furoyl)-4(5)-(2-furanyl)-1H-imidazole (FFI), was reported by Pongor et al. (Pongor et al., 1984). Wolff and co-workers reported the slow oxidative degradation of monosaccharides under physiological conditions to form the corresponding ɑ-oxoaldehyde and hydrogen peroxide. This process was called monosaccharide autoxidation (Wolff et al., 1984).
In 1986, Cerami first used the term advanced glycation endproducts (AGEs) to refer to “brown fluorescent pigments which crosslink proteins” formed from the degradation of fructosamine (Cerami, 1986). Brownlee et al. reported increased collagen cross-linking in the arterial walls of diabetic rats and associated fluorescence characteristic of AGE compounds. The nucleophilic hydrazine derivative, aminoguanidine (PimagedineTM), inhibited the formation of AGEs and diabetes-induced protein crosslinking (Brownlee et al., 1986). Ahmed et al. also reported the formation of Nɛ-carboxymethyl-lysine (CML) from the oxidative degradation of glycated proteins and also noticed that these two compounds were present in human urinary metabolite (Ahmed et al., 1986). CML was later found to be formed by the reaction of lysine residues with ascorbate and glyoxal formed in lipid peroxidation and is now one of the most studied AGEs in glycation research (Thorpe and Baynes, 2002).
In 1988, Thornalley presented evidence linking hyperglycaemia in diabetes mellitus with increased flux of formation and concentration of MG (Thornalley, 1988). Glycation by MG was later to be proposed as one of the key pathways driving the development of vascular complications of diabetes (Brownlee, 2001).
In 1989, Monnier and Sell isolated an acid stable fluorescent compound from collagen – “pentosidine”. It was a crosslink formed from a pentose moiety with lysine and arginine residues and is a further highly studied AGE (Sell and Monnier, 1989). In 1991, the enzymatic metabolism of fructosamine adducts was also discovered by Horiuchi and Kyurokawa - fructosyl-amino acid oxidase from bacteria catalysed the conversion of fructosyl-amino acids to the free amino acid, glucosone and hydrogen peroxide (Horiuchi and Kurokawa, 1991).
In 1992, Schmidt and co-workers identified an AGE receptor protein from bovine lung endothelial cells – later shown to have a sequence mass of 42 kDa (Schmidt et al., 1992). This was the Receptor for Advanced Glycosylation Endproducts (RAGE) and is the best characterised of the AGE receptors.
Over the period 1994 - 1998 the inhibitor of AGE formation PimagedineTM was evaluated in clinical trials for the prevention of overt nephropathy in clinical diabetes (ACTION I and ACTION II). The trials were eventually terminated due to safety concerns and apparent lack of efficacy (Thornalley, 2003c).
In 1996, Cerami and co-workers introduced the concept of “AGE breakers” – compounds that may cleave glycation derived crosslinks and reverse one of the damaging effects of glycation associated with ageing and disease. The prototype compound was N-phenacylthiazolium bromide (PTB) (Vasan et al., 1996). Unfortunately it and related analogues had stability problems (Price et al., 2001,Thornalley and Minhas, 1999).
In 1997, Shipanova et al. discovered argpyrimidine – a major fluorescence AGE formed from MG (Shipanova et al., 1997). Vlassara and co-workers used the term “glycotoxins” to refer to highly reactive AGE intermediates and initiated a series of on-going studies assessing the physiological effects of AGEs in the diet on health (Koschinsky et al., 1997).
In 2000, Delpierre et al. discovered fructosamine 3-kinase which catalyses the phosphorylation of fructosamine and fructosamine residues in proteins leading to the de-glycation and repair of early glycated proteins (Delpierre et al., 2000). A time-line diagram of glycation research development is given in Figure 3.
Figure 3. Timeline showing key develops in glycation research from 1900 - 2000. Abbreviation: GA3P, glyceraldehyde-3-phosphate.
Since 2000 there has been a marked expansion of glycation-related research – particularly with application of proteomics to detection and quantitation of glycated proteins and glycation free adducts, cell signalling studies in response to RAGE and other response, functional genomics of components of enzymatic defence against glycation (fructosamine 3-kinase, glyoxalase 1 and aldoketo reductases), therapeutics development, food processing and other applications.
Importance of protein glycation
In biology protein glycation is implicated in the low-level endogenous damage to the proteome with the ubiquitous presence of an enzymatic defence akin to that of antioxidants to counter it. The enzymatic defence against glycation consists of enzymes that repair early glycated proteins and prevent glycation by metabolising dicarbonyl glycating agents: fructosamine 3-kinase (Delpierre et al., 2000), fructosamine 3-kinase-related protein (Delplanque et al., 2004) and others; and glyoxalase 1 (Thornalley, 2003b) and aldoketo reductases 1A4, 1B1 and 1B3 (aldose reductase) (Baba et al., 2009). The imbalance of glycating agents and enzymatic defence against glycation in favour of glycating agents has been termed carbonyl stress and is thought to contribute to ageing and disease (Miyata et al., 1999,Morcos et al., 2008,Xue et al., 2008). Emerging evidence of a chaperone function of lens crystallins (Gangadhariah et al., 2010) and a regulatory decrease in the enzymatic defence against glycation in inflammatory signalling (Thornalley, 2007) is suggesting a beneficial functional role of protein glycation in some instances.
In the clinical setting, increased formation and steady-state accumulation of glycated proteins has been linked to disease development and progression – particularly diabetes and its vascular complications (Ahmed et al., 2005a,McCance et al., 1993), renal failure (Agalou et al., 2005,Miyata et al., 2001), cardiovascular disease and Alzheimer’s disease (Ahmed et al., 2004a,Chen et al., 2004,Rabbani et al., 2010). Involvement in arthritis and cirrhosis (Ahmed et al., 2004b,Ahmed et al., 2006), mood affective disorders – anxiety and Schizophrenia (Arai et al., 2010,Hambsch et al., 2010), Parkinson’s disease (Kurz et al., 2010) and other disease is emerging.
In food and nutrition, glycation is increased in culinary processing – particularly during heating and alkaline-treated bakery products (Ahmed et al., 2005b,Henle, 2001). Glycation may impair nutritional quality, influence intestinal microflora, post-prandial vascular dysfunction, glucose tolerance and obesity (Birlouez-Aragon et al., 2010,Tuohy et al., 2006). The contribution of glycation adducts in proteins in food to total physiological exposure to glycation adducts and pathological effect remains controversial (Henle, 2007,Uribarri et al., 2010). Glycation adduct contents of food, pre-absorptive processing and metabolic transit of protein glycation adducts (bioavailability, clearance and residence time in the body) and functional effects remain critical issues for further investigation.
In pharmacology and toxicology, multidrug resistance associated with overexpression of glyoxalase 1 has suggested a role of dicarbonyl glycation in the mechanism of action of cytotoxic antitumour agents (Thornalley, 2003a). Fortunately tumours with overexpression of glyoxalase – now know to be linked to Glo1 gene amplification (Santarius et al., 2010) – are sensitive to glyoxalase 1 inhibitor therapy (Sakamoto et al., 2001,Thornalley et al., 1996). Drug development to increase dicarbonyl glycation by inhibition of glyoxalase 1 will likely find use in overcome tumour multidrug resistance and drug and dietary supplement development to decrease glycation will find use as disease therapeutics and prophylaxis for healthy ageing (Morcos et al., 2008).
Technological processing of medical products such as dialyse fluids for renal replacement therapy (Linden et al., 2002,Schalkwijk et al., 1999) and food and beverages such as sweetened cola drinks involve formation and exposure to dicarbonyl glycating agents (Lo et al., 2008). Formation of crosslinking in proteins in tanning and desired browning of skin in lotions also involves glycation (Avery and Bailey, 2008,Petersen et al., 2004). Further development will optimise the levels of glycating agents and type and levels of glycation adducts for technological application.
In the clinical setting, increased formation and steady-state accumulation of glycated proteins has been linked to disease development and progression – particularly diabetes and its vascular complications (Ahmed et al., 2005a,McCance et al., 1993), renal failure (Agalou et al., 2005,Miyata et al., 2001), cardiovascular disease and Alzheimer’s disease (Ahmed et al., 2004a,Chen et al., 2004,Rabbani et al., 2010). Involvement in arthritis and cirrhosis (Ahmed et al., 2004b,Ahmed et al., 2006), mood affective disorders – anxiety and Schizophrenia (Arai et al., 2010,Hambsch et al., 2010), Parkinson’s disease (Kurz et al., 2010) and other disease is emerging.
In food and nutrition, glycation is increased in culinary processing – particularly during heating and alkaline-treated bakery products (Ahmed et al., 2005b,Henle, 2001). Glycation may impair nutritional quality, influence intestinal microflora, post-prandial vascular dysfunction, glucose tolerance and obesity (Birlouez-Aragon et al., 2010,Tuohy et al., 2006). The contribution of glycation adducts in proteins in food to total physiological exposure to glycation adducts and pathological effect remains controversial (Henle, 2007,Uribarri et al., 2010). Glycation adduct contents of food, pre-absorptive processing and metabolic transit of protein glycation adducts (bioavailability, clearance and residence time in the body) and functional effects remain critical issues for further investigation.
In pharmacology and toxicology, multidrug resistance associated with overexpression of glyoxalase 1 has suggested a role of dicarbonyl glycation in the mechanism of action of cytotoxic antitumour agents (Thornalley, 2003a). Fortunately tumours with overexpression of glyoxalase – now know to be linked to Glo1 gene amplification (Santarius et al., 2010) – are sensitive to glyoxalase 1 inhibitor therapy (Sakamoto et al., 2001,Thornalley et al., 1996). Drug development to increase dicarbonyl glycation by inhibition of glyoxalase 1 will likely find use in overcome tumour multidrug resistance and drug and dietary supplement development to decrease glycation will find use as disease therapeutics and prophylaxis for healthy ageing (Morcos et al., 2008).
Technological processing of medical products such as dialyse fluids for renal replacement therapy (Linden et al., 2002,Schalkwijk et al., 1999) and food and beverages such as sweetened cola drinks involve formation and exposure to dicarbonyl glycating agents (Lo et al., 2008). Formation of crosslinking in proteins in tanning and desired browning of skin in lotions also involves glycation (Avery and Bailey, 2008,Petersen et al., 2004). Further development will optimise the levels of glycating agents and type and levels of glycation adducts for technological application.
The future of glycation research
Glycation research poses great challenges to scientists. The extent of glycation of proteins in physiological samples is typically low and the range of adducts diverse in physiological systems. In culinary processed proteins of foods, the content of glycation adducts is high. AGE content of foods is often difficult to analyse intact and resistant to release without decomposition for analysis. Risk of formation of glycation adducts and some of their precursors during pre-analytic processing is nearly always significant and overestimation of analytes is often possible. The physiological effects of protein glycation are diverse, intricate and complex.
The history of glycation research continues to be written. Some long-held views on glycation hold good and some have been revised in recent years. This is how scientific research advances: through inspired and ground-breaking hypothesis with amendment or rejection of hypotheses which fail to be supported by experimental evidence. One thing is certain: glycation research has much of importance still to deliver.
From Rabbani, N. and Thornalley, P. J. (2012) Glycation research in Amino Acids: a place to call home. Amino Acids. 42, 1087-1096, with minor modification.
Naila Rabbani and Paul J Thornalley, 30th December 2019.
The history of glycation research continues to be written. Some long-held views on glycation hold good and some have been revised in recent years. This is how scientific research advances: through inspired and ground-breaking hypothesis with amendment or rejection of hypotheses which fail to be supported by experimental evidence. One thing is certain: glycation research has much of importance still to deliver.
From Rabbani, N. and Thornalley, P. J. (2012) Glycation research in Amino Acids: a place to call home. Amino Acids. 42, 1087-1096, with minor modification.
Naila Rabbani and Paul J Thornalley, 30th December 2019.
References
- Agalou S, Ahmed N, Babaei-Jadidi R, Dawnay A, Thornalley PJ (2005) Profound mishandling of protein glycation degradation products in uremia and dialysis. J Am Soc Nephrol 16: 1471-1485
- Ahmed MU, Thorpe SR, Baynes JW (1986) Identification of Ne-carboxymethyl-lysine as a degradation product of fructoselysine in glycated protein. J Biol Chem 261: 4889-4894
- Ahmed N, Ahmed U, Thornalley PJ, Hager K, Fleischer GA, Munch G (2004a) Protein glycation, oxidation and nitration marker residues and free adducts of cerebrospinal fluid in Alzheimer's disease and link to cognitive impairment. J Neurochem 92: 255-263
- Ahmed N, Ahmed U, Thornalley PJ, Watts R, Tarr J, Haigh R, and Winyard P. (2006) Profound increase in proteolytic products of glycated and oxidised proteins in synovial fluid and plasma in osteoarthritis and rheumatoid arthritis, corrected by TNF-α antibody therapy in rheumatoid arthritis. Rheumatology 45, Suppl. 1: i53.
- Ahmed N, Argirov OK, Minhas HS, Cordeiro CA, Thornalley PJ (2002) Assay of advanced glycation endproducts (AGEs): surveying AGEs by chromatographic assay with derivatisation by aminoquinolyl-N-hydroxysuccimidyl-carbamate and application to Ne-carboxymethyl-lysine- and Ne-(1-carboxyethyl)lysine-modified albumin. Biochem J 364: 1-14
- Ahmed N, Babaei-Jadidi R, Howell SK, Beisswenger PJ, Thornalley PJ (2005a) Degradation products of proteins damaged by glycation, oxidation and nitration in clinical type 1 diabetes. Diabetologia 48: 1590-1603
- Ahmed N, Mirshekar-Syahkal B, Kennish L, Karachalias N, Babaei-Jadidi R, Thornalley PJ (2005b) Assay of advanced glycation endproducts in selected beverages and food by liquid chromatography with tandem mass spectrometric detection. Molec Nutrit & Food Res 49: 691-699
- Ahmed N, Thornalley PJ, Luthen R, Haussinger D, Sebekova K, Schinzel R, Voelker W, Heidland A (2004b) Processing of protein glycation, oxidation and nitrosation adducts in the liver and the effect of cirrhosis. J Hepatol 41: 913-919
- Allen DW, Schroeder WA, Balog J (1958) Observations on the Chromatographic Heterogeneity of Normal Adult and Fetal Human Hemoglobin: A Study of the Effects of Crystallization and Chromatography on the Heterogeneity and Isoleucine Content. J Am Chem Soc 80: 1628-1634
- Amadori M (1929a) The condensation product of glucose and p-anisidine. Atti Reale Accad Nazl Lincei 9: 226-230
- Amadori M (1929b) The product of the condensation of glucose and p-phenetidine. Atti Reale Accad Nazl Lincei 9: 68-73
- Anet EFLJ (1960) Degradation of carbohydrates. I. Isolation of 3-deoxyhexosones. Australian J Chem 13: 396-403
- Arai M, Yuzawa H, Nohara I, Ohnishi T, Obata N, Iwayama Y, Haga S, Toyota T, Ujike H, Arai M, Ichikawa T, Nishida A, Tanaka Y, Furukawa A, Aikawa Y, Kuroda O, Niizato K, Izawa R, Nakamura K, Mori N, Matsuzawa D, Hashimoto K, Iyo M, Sora I, Matsushita M, Okazaki Y, Yoshikawa T, Miyata T, Itokawa M (2010) Enhanced Carbonyl Stress in a Subpopulation of Schizophrenia. Arch Gen Psychiatry 67: 589-597
- Avery N, Bailey A (2008) Restraining cross-links responsible for the mechanical properties of collagen fibers: Natural and artificial. Collagen: Structure and Mechanics 81-110
- Baba SP, Barski OA, Ahmed Y, O'Toole TE, Conklin DJ, Bhatnagar A, Srivastava S (2009) Reductive Metabolism of AGE Precursors: A Metabolic Route for Preventing AGE Accumulation in Cardiovascular Tissue. Diabetes 58: 2486-2497
- Baynes JW (1991) Role of oxidative stress in the development of complications in diabetes. Diabetes 40: 405-412
- Baynes JW, Thorpe SR (2000) Glycoxidation and lipoxidation in atherogenesis. Free Radical Biology & Medicine 28: 1708-1716
- Benedict SR (1909) A reagent for the detection of reducing sugars . J Biol Chem 5: 485-487
- Birlouez-Aragon I, Saavedra G, Tessier FJ, Galinier A, Ait-Ameur L, Lacoste F, Niamba CN, Alt N, Somoza V, Lecerf JM (2010) A diet based on high-heat-treated foods promotes risk factors for diabetes mellitus and cardiovascular diseases. Am J Clin Nutr 91: 1220-1226
- Bonsignore A, Leoncini G, Siri A, Ricci D (1973) Kinetic behaviour of glyceraldehyde 3-phosphate conversion into methylglyoxal. Ital J Biochem 22: 131-140
- Bookchin RM, Gallop PM (1968) Structure of hemoglobin A1c: nature of the N-terminal b-chain blocking group. Biochem Biophys Res Com 32: 86-93
- Brownlee M (2001) Biochemistry and molecular cell biology of diabetic complications. Nature 414: 813-820
- Brownlee M, Vlassara H, Kooney A, Ulrich P, Cerami A (1986) Aminoguanidine prevents diabetes-induced arterial wall protein cross-linking. Science 232: 1629-1632
- Bunn HF, Haney DN, Gabbay KH, Gallop PM (1975) Further identification of the nature and linkage of the carbohydrate in hemoglobin A1c. Biochemical and Biophysical Research Communications 67: 103-109
- Cerami A (1986) Aging of proteins and nucleic acids: what is the role of glucose? TIBS 11: 311-314
- Chen F, Wollmer MA, Hoerndli F, Munch G, Kuhla B, Rogaev EI, Tsolaki M, Papassotiropoulos A, Gotz J (2004) Role for glyoxalase I in Alzheimer's disease. Proc Natl Acad Sci USA 101: 7687-7692
- Dakin HD, Dudley HW (1913) An enzyme concerned with the formation of hydroxy acids from ketonic aldehydes. J Biol Chem 14: 155-157
- Delpierre G, Rider MH, Collard F, Stroobant V, Vanstapel F, Santos H, Van Schaftingen E (2000) Identification, cloning, and heterologous expression of a mammalian fructosamine-3-kinase. Diabetes 49: 1627-1634
- Delplanque J, Delpierre G, Opperdoes FR, Van Schaftingen E (2004) Tissue Distribution and Evolution of Fructosamine 3-Kinase and Fructosamine 3-Kinase-related Protein. Journal of Biological Chemistry 279: 46606-46613
- Fu M-X, Requena JR, Jenkins AJ, Lyons TJ, Baynes JW, Thorpe SR (1996) The advanced glycation end product, Ne-(carboxymethyl)lysine, is a product of both lipid peroxidation and glycoxidation reactions. J Biol Chem 271: 9982-9986
- Gangadhariah MH, Wang BL, Linetsky M, Henning C, Spanneberg R, Glomb MA, Nagaraj RH (2010) Hydroimidazolone modification of human alpha A-crystallin: Effect on the chaperone function and protein refolding ability. Biochimica et Biophysica Acta-Molecular Basis of Disease 1802: 432-441
- Hambsch B, Chen BG, Brenndorfer J, Meyer M, Avrabos C, Maccarrone G, Liu RH, Eder M, Turck CW, Landgraf R (2010) Methylglyoxal-mediated anxiolysis involves increased protein modification and elevated expression of glyoxalase 1 in the brain. Journal of Neurochemistry 113: 1240-1251
- Hayashi T, Namiki M (1980) Formation of two-carbon sugar fragments at an early stage of the browning reaction of sugar and amine. Agric Biol Chem 44: 2575-2580
- Henle T (2001) A food chemist's view of advanced glycation end-products. Perit Dial Int 21: S125-S130
- Henle T (2007) Dietary advanced glycation end products - a risk to human health? A call for an interdisciplinary debate. Molecular Nutrition & Food Research 51: 1075-1078
- Henle T, Walter A, Haebner R, Klostermeryer H (1994) Detection and identification of a protein-bound imidazolone resulting from the reaction of arginine residues and methylglyoxal. Z Lebensm Unters Forsch 199: 55-58
- Hodge JE (1953) Dehydrated foods: chemistry of browning reactions in model systems. J Agric Food Chem 1: 928-943
- Hodge JE (1955) The Amadori rearrangement. Adv Carbohydr Chem 10: 169-205
- Horiuchi T, Kurokawa T (1991) Purification and properties of fructosylamine oxidase from Aspergillus sp. 1005. Agric Biol Chem 55: 333-338
- Kato H (1960) Studies on browning reactions between sugars and amino compounds. V. Isolation and characterisation of new carbonyl compounds, 3-deoxyglucosones formed from N-glycosides and their significance for browning reaction. Bull Agric Chem Soc Japan 24: 1-12
- Kato H, Hayase F, Shin DB, Oimomi M, Baba S (1989) 3-Deoxyglucosone, an intermediate product of the Maillard reaction. In The Maillard reaction in aging, diabetes and nutrition, pp 69-84. Alan R. Liss inc.:
- Kato H, Yamamoto M, Fujimaki M (1969) Mechanisms of browning degradation of D-fructose in special comparison with D-glucose-glycine reaction. Agric Biol Chem 33: 939-948
- Kawamura S (1983) 70 Years of the Maillard-Reaction. Acs Symposium Series 215: 3-18
- Koenig RJ, Peterson CM, Jones RL, Saudek C, Lehrman M, Cerami A (1976) A correlation of glucose regulation and Hemoglobin A1c in diabetes mellitus. New Engl J Med 295: 417-420
- Koschinsky T, He C-J, Mitsuhashi T, Bucala R, Liu C, Buenting C, Heitman K, Vlassara H (1997) Orally absorbed reactive glycation products (glycotoxins): an environmental risk factor in diabetic nephropathy. Proc Natl Acad Sci USA 94: 6474-6479
- Kuhn R, Dansi A (1936) A molecular rearrangement of N-glucosides. Ber 69B: 1745-1754
- Kuhn R, Weygand F (1937) The Amadori rearrangement. Ber 70B: 769-772
- Kurz A, Rabbani N, Walter M, Bonin M, Thornalley PJ, Auburger G, and Gispert S. (2011) Alpha-synuclein deficiency leads to increased glyoxalase I expression and glycation stress. Cell and Molecular Life Sci 68: 721-733.
- Linden T, Cohen A, Deppisch R, Kjellstrand P, Wieslander A (2002) 3,4-Dideoxyglucosone-3-ene (3,4-DGE): A cytotoxic glucose degradation product in fluids for peritoneal dialysis. Kidney Int 62: 697-703
- Ling AR (1908) Malting. J Inst Brewing 14: 521
- Lis H, Sharon N (1993) Protein Glycosylation - Structural and Functional-Aspects. European Journal of Biochemistry 218: 1-27
- Lo CY, Li S, Wang Y, Tan D, Pan MH, Sang S, Ho CT (2008) Reactive dicarbonyl compounds and 5-(hydroxymethyl)-2-furfural in carbonated beverages containing high fructose corn syrup. Food Chemistry 107: 1099-1105
- Maillard LC (1912a) Action des acides amines sur les sucres: Formation des melanoidines par voie methodique. Compt Rend Hebd Seances Acad Sci 154: 66-68
- Maillard LC (1912b) Formation d'humus et de combustibles mineraux sans intervention de l'oxygene atmospherique des microorganismes, des hautes temperatures, ou des fortes pressions. Compt Rend Hebd Seances Acad Sci 155: 1554-1556
- McCance DR, Dyer DG, Dunn JA, Baiue KE, Thorpe SR, Baynes JW, Lyons TJ (1993) Maillard reaction products and their relation to complications in insulin-dependent diabetes mellitus. J Clin Invest 91: 2470-2478
- Miyata T, van Ypersele de Strihou C, Imasawa T, Yoshino A, Ueda Y, Ogura H, Kominami K, Onogi H, Inagi R, Nangaku M, Kurokawa K (2001) Glyoxalase I deficiency is associated with an unusual level of advanced glycation end products in a hemodialysis patient. Kidney Internat 60: 2351-2359
- Miyata T, van Ypersele de Strihou C, Kurokawa H, Baynes JW (1999) Alterations in nonenzymatic biochemistry in uraemia: origin and significance of "carbonyl stress" in long-term uremic complications. Kidney Int 55: 389-399
- Morcos M, Du X, Pfisterer F, Hutter H, Sayed AAR, Thornalley P, Ahmed N, Baynes J, Thorpe S, Kukudov G, Schlotterer A, Bozorgmehr F, El Baki RA, Stern D, Moehrlen F, Ibrahim Y, Oikonomou D, Hamann A, Becker C, Zeier M, Schwenger V, Miftari N, Humpert P, Hammes HP, Buechler M, Bierhaus A, Brownlee M, Nawroth PP (2008) Glyoxalase-1 prevents mitochondrial protein modification and enhances lifespan in Caenorhabditis elegans. Aging Cell 7: 260-269
- Nakayama T, Hayase F, Kato H (1980) Formation of Ne-(2-formyl-5-hydroxy-methyl-pyrrol-1-yl)-L- norleucine in the Maillard reaction between D-glucose and L-lysine. Agric Biol Chem 44: 1201-1202
- Neuberg C (1913) The destruction of lactic aldehyde and methylglyoxal by animal organs. Biochem Z 49: 502-506
- Petersen AB, Wulf HC, Gniadecki R, Gajkowska B (2004) Dihydroxyacetone, the active browning ingredient in sunless tanning lotions, induces DNA damage, cell-cycle block and apoptosis in cultured HaCaT keratinocytes. Mutation Research-Genetic Toxicology and Environmental Mutagenesis 560: 173-186
- Pongor S, Ulrich PC, Benesath FA, Cerami A (1984) Aging of protiens: isolation and identification of a fluorescent chromophore from the reaction of polypeptides with glucose. Proc Natl Acad Sci USA 81: 2684-2688
- Price DL, Rhett PM, Thorpe SR, Baynes JW (2001) Chelating activity of advanced glycation end-product inhibitors. J Biol Chem 276: 48967-48972
- Rabbani N, Sebekova K, Sebekova K, Jr., Heidland A, Thornalley PJ (2007) Protein glycation, oxidation and nitration free adduct accumulation after bilateral nephrectomy and ureteral ligation. Kidney Internat 72: 1113-1121
- Rabbani N, Varma Chittari M, Bodmer CW, Zehnder D, Ceriello A, Thornalley PJ (2010) Increased glycation and oxidative damage to apolipoprotein B100 of LDL in patients with type 2 diabetes and effect of metformin. Diabetes 59: 1038-1045
- Rahbar S (1968) An abnormal hemoglobin in red cells of diabetics. Clin Chim Acta 22: 296-298
- Sakamoto H, Mashima T, Sato S, Hashimoto Y, Yamori T, Tsuruo T (2001) Selective activation of apoptosis program by S-p-bromobenzylglutathione cyclopentyl diester in glyoxalase I-overexpressing human lung cancer cells. Clin Cancer Res 7: 2513-2518
- Santarius T, Bignell GR, Greenan CD, Widaa S, Chen L, Mahoney CL, Butler A, Edkins S, Waris S, Thornalley PJ, Futreal PA, Stratton MR (2010) GLO1 - A novel amplified gene in human cancer. Genes, Chromsomes and Cancer 49: 711-725
- Schalkwijk CG, Posthuma N, ten Brink HJ, ter Wee PM, Teerlink T (1999) Induction of 1,2-dicarbonyl compounds, intermediates in the formation of advanced glycation end-products, during heat-sterilization of glucose-based peritoneal dialysis fluids. Peritoneal Dialysis Internat 19: 325-333
- Schmidt A-M, Vianna M, Gerlach M, Brett J, Ryan J, Kao J, Esposito C, Hegarty H, Hurley W, Clauss M, Wang F, Pan YE, Tsang TE, Stern D (1992) Isolation and characterization of two binding proteins for advanced glycosylation endproducts from bovine lung which are present on the endothelial cell surface. J Biol Chem 267: 14987-14997
- Sell DR, Monnier VM (1989) Structure elucidation of a senescence crosslink from human extracellular matrix. Implication of pentoses in the aging process. J Biol Chem 264: 21597-21602
- Sharon N (1986) IUPAC-IUB Joint Commission on Biochemical Nomenclature (Jcbn) - Nomenclature of Glycoproteins, Glycopeptides and Peptidoglycans - Jcbn Recommendations 1985. Glycoconjugate Journal 3: 123-134
- Shipanova IN, Glomb MA, Nagaraj RH (1997) Protein modification by methylglyoxal: chemical nature and synthetic mechanism of a major fluorescent adduct. Arch Biochem Biophys 344: 29-36
- Takahashi K (1977) Further studies on the reactions of phenylglyoxal and related reagents with proteins. Biochem J 81: 403-414
- Thornalley PJ (1988) Modification of the glyoxalase system in human red blood cells by glucose in vitro. Biochem J 254: 751-755
- Thornalley PJ (2003a) Protecting the genome: defence against nucleotide glycation and emerging role of glyoxalase I over expression in multidrug resistance in cancer chemotherapy. Biochem Soc Trans 31: 1372-1377
- Thornalley PJ (2003b) The enzymatic defence against glycation in health, disease and therapeutics: a symposium to examine the concept. Biochem Soc Trans 31: 1343-1348
- Thornalley PJ (2003c) Use of aminoguanidine (Pimagedine) to prevent the formation of advanced glycation endproducts. Arch Biochem Biophys 419: 31-40
- Thornalley PJ (2007) Dietary AGEs and ALEs and risk to human health by their interaction with the receptor for advanced glycation endproducts (RAGE) - an introduction. Molec Nutrit and Food Res 51: 1107-1110
- Thornalley PJ, Battah S, Ahmed N, Karachalias N, Agalou S, Babaei-Jadidi R, Dawnay A (2003) Quantitative screening of advanced glycation endproducts in cellular and extracellular proteins by tandem mass spectrometry. Biochem J 375: 581-592
- Thornalley PJ, Edwards LG, Kang Y, Wyatt C, Davies N, Ladan MJ, Double J (1996) Antitumour activity of S-p-bromobenzylglutathione cyclopentyl diester in vitro and in vivo. Inhibition of glyoxalase I and induction of apoptosis. Biochem Pharmacol 51: 1365-1372
- Thornalley PJ, Minhas HS (1999) Rapid hydrolysis and slow a,b-dicarbonyl cleavage of an agent proposed to cleave glucose-derived protein cross-links. Biochem Pharmacol 57: 303-307
- Thorpe SR, Baynes JW (2002) CML: a brief history. Maillard Reaction in Food Chemistry and Medical Science: Update for the Postgenomic Era 1245: 91-99
- Tuohy KM, Hinton DJS, Davies SJ, Crabbe MJC, Gibson GR, Ames JM (2006) Metabolism of Maillard reaction products by the human gut microbiota - implications for health. Molecular Nutrition & Food Research 50: 847-857
- Uribarri J, Woodruff S, Goodman S, Cai WJ, Chen X, Pyzik R, Yong A, Striker GE, Vlassara H (2010) Advanced Glycation End Products in Foods and a Practical Guide to Their Reduction in the Diet. Journal of the American Dietetic Association 110: 911-916
- Vasan S, Zhang X, Kapurniotu A, Bernhagen J, Teichberg S, Basgen J, Wagle D, Shih D, Terlecky I, Bucala R, Cerami A, Egan J, Ulrich P (1996) An agent cleaving glucose-derived protein crosslinks in vitro and in vivo. Nature 382: 275-278
- Wolff SP, Crabbe MJC, Thornalley PJ (1984) The autoxidation of a-hydroxyketones. Experientia 40: 244-246
- Xue M, AntonySunil A, Rabbani N, Thornalley PJ (2008) Protein damage by glycation, oxidation and nitration in the ageing process. Advances in quantitation of protein damage and the emerging importance of decline in enzymatic defences as the ageing phenotype develops. In Redox Metabolism and Longevity Relationships in Animals and Plants, Foyer CH, Faraghar R, Thornalley PJ (eds) pp 227-265. Garland Science: London